Replace Word for
Medical Writing
Studia is the medical writing platform that connects regulatory guidelines and learnings from past clinical studies – helping you write faster but, more importantly, better. We help draft and check Protocols, ICFs, IBs,…
You get compliance at the first iteration.
Trusted by medical writers at sponsors, CROs, and independent practices worldwide
Writing regulatory documents
in Word is broken
Weeks spent cross-checking guidelines
You toggle between Word, PDFs, and browser tabs to check ICH, EMA/FDA, CTR/MDR, and other local guidelines. Every citation is manual. Every format requirement is checked by hand.
Inconsistencies that slip through to submission
No automated compliance checking. Errors get caught late — or trigger amendments that cost hundreds of thousands and delay patient access to treatments.
Knowledge scattered across silos
Past protocols in shared drives. ClinicalTrials.gov and EU CTIS in separate tabs. PubMed in another. Internal SOPs and Meeting Notes somewhere else. Nothing connected, nothing searchable.
A purpose-built writing environment for clinical documents
Studia brings GCP and regulatory guidelines, past clinical studies data, and AI into one editor — so you write, collaborate, check, validate, and export compliant documents without leaving the platform.
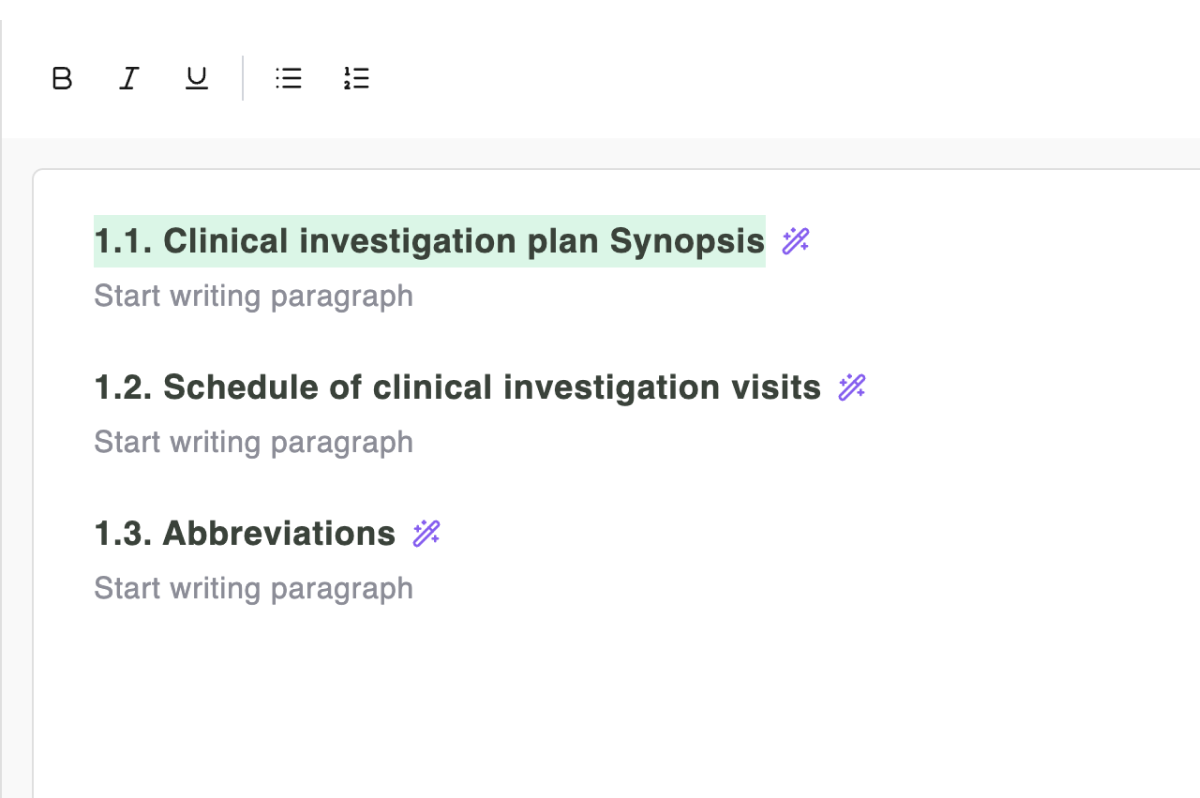
Write in a familiar editor
A Word-like writing experience built for regulatory documents. No learning curve — just open a template and start writing. Compliance is built into every paragraph.
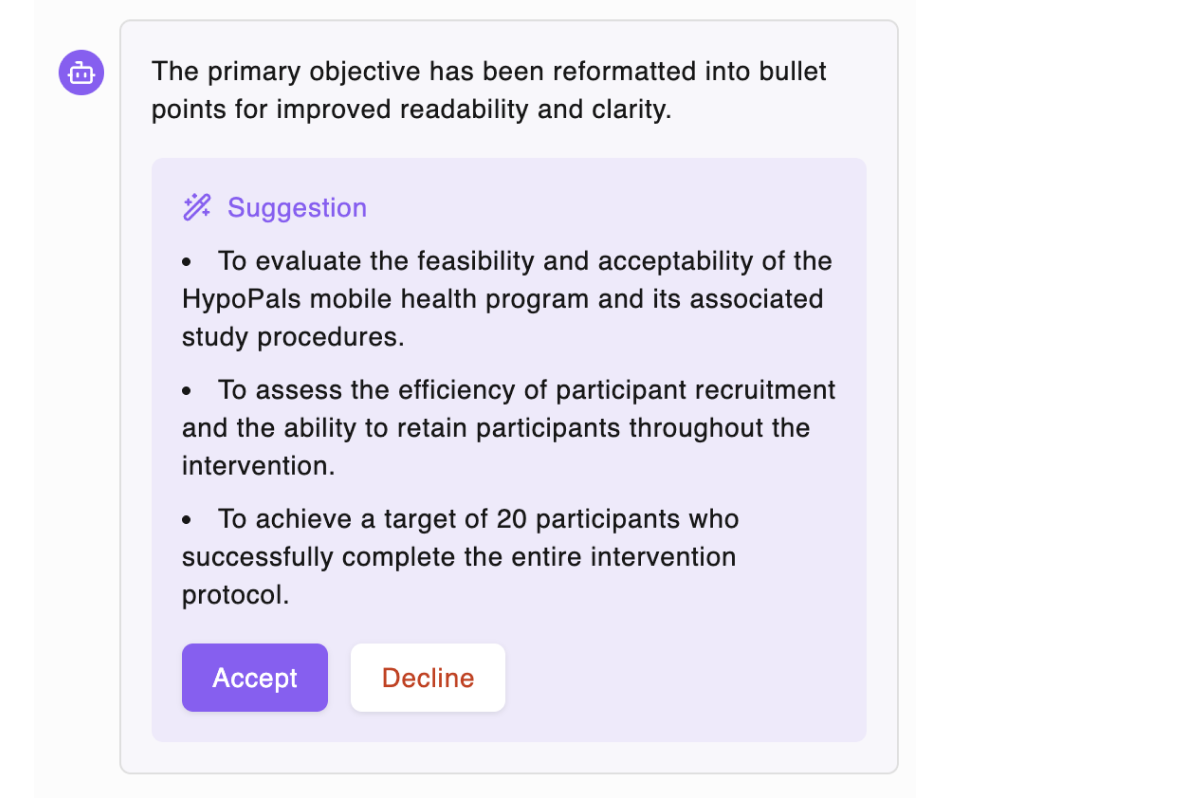
AI generates compliant first drafts
Generate content grounded in regulatory guidelines and historical trial data — not hallucinated text. Review, edit, and approve. You stay in control.
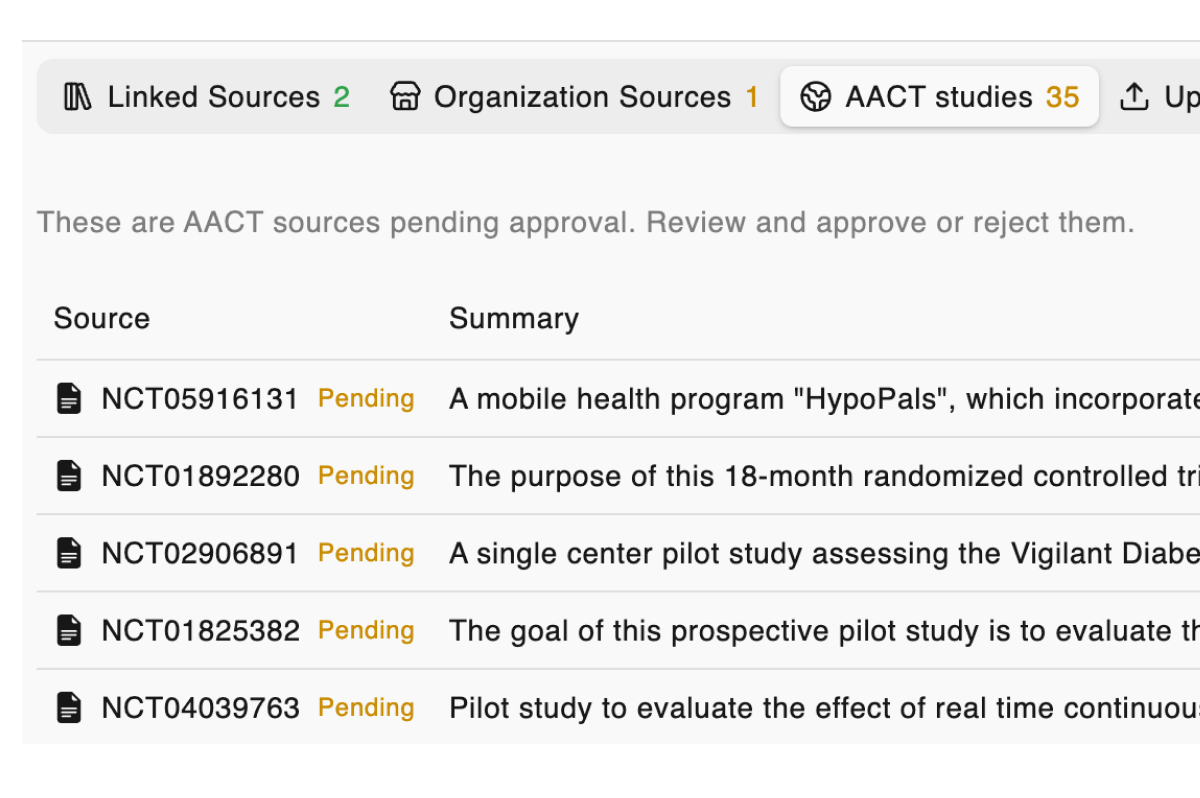
All your sources, connected
ClinicalTrials.gov, EU CTIS, PubMed, and your internal documents — linked, searchable, and cited automatically. No more toggling between tabs.
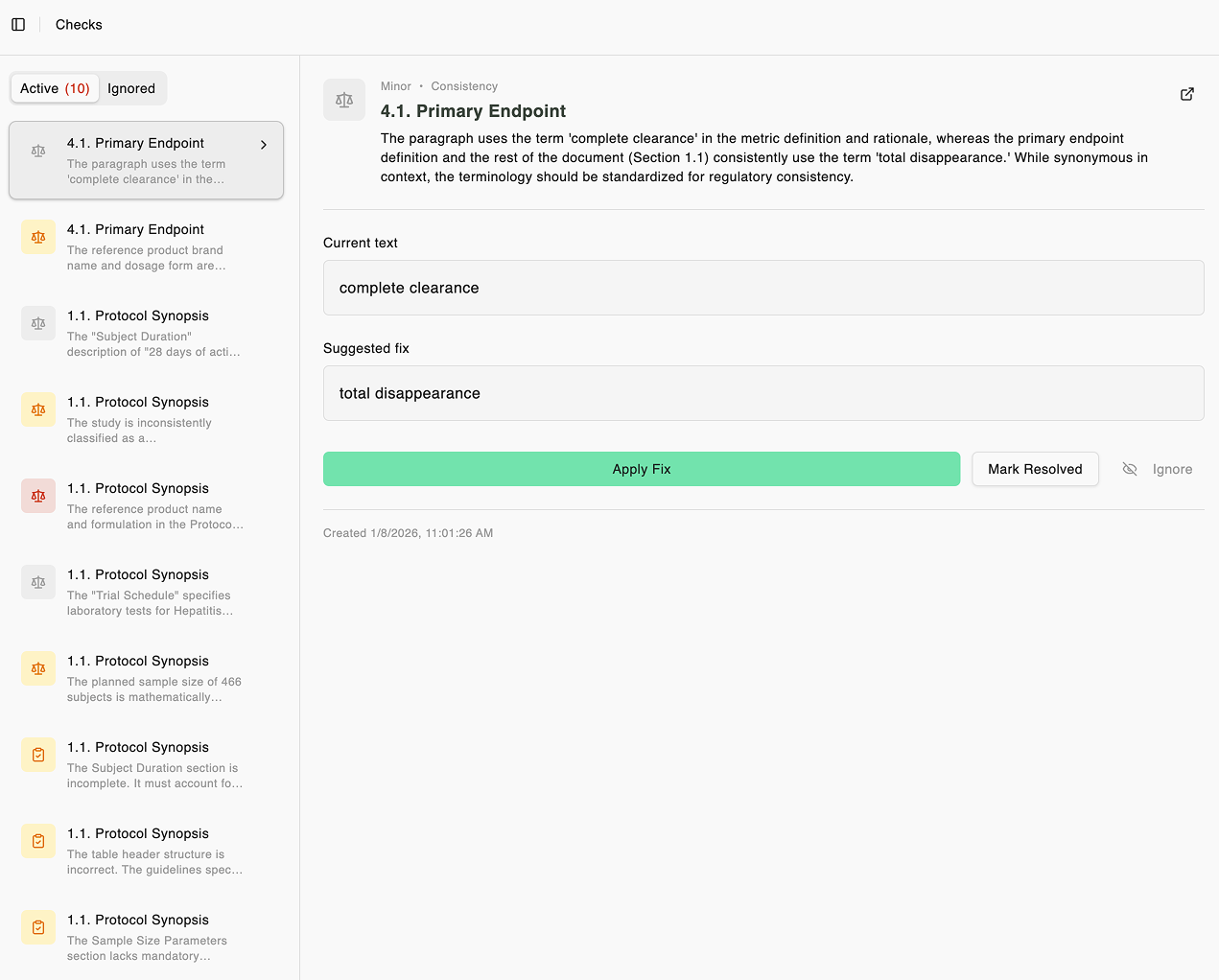
Automatic quality checks before submission
Compliance verification, redundancy detection, and inconsistency checks — all running automatically. Catch guideline violations and contradictions before reviewers do.
Version control
Track every change with full document history and revision comparison.
Real-time collaboration
Work on the same document with your team simultaneously.
Word & PDF export
Export submission-ready documents in the format regulators expect.
Trusted by medical writers worldwide
Studia has displayed foundational steps as a next-generation authoring environment for clinical protocol development. Its intuitive interface and ability to generate logical, compliant clinical text represent a clear advancement in operational efficiency.
Studia truly transforms how researchers develop clinical trial protocols. Its AI-driven precision, intelligent referencing from sources like ClinicalTrials.gov, and flexible editing features give researchers complete control over their work while saving an enormous amount of time.
A structured, user-friendly environment for creating clinical investigation plans with AI support and guideline integration. Source linking, export, and historical features support regulatory compliance and audit readiness.
The built-in AI assistant made my writing more accurate and cleaner, and helped me move through sections much faster. It brought in relevant studies and background details, saving me a lot of time searching.
Meet Our Team
Experts in medical writing, AI, and healthcare technology
Our Mission
We are dedicated to helping patients worldwide by accelerating clinical studies.
We streamline medical writing and regulatory documentation to bring life-saving drugs and medical devices to market faster. Every day saved in the approval process means more patients can access innovative treatments sooner.


Ready to replace Word?
See how Studia can cut your document creation time in half. Schedule a demo or send us a message.








